How does ice cream salt work?
How does ice cream salt work?
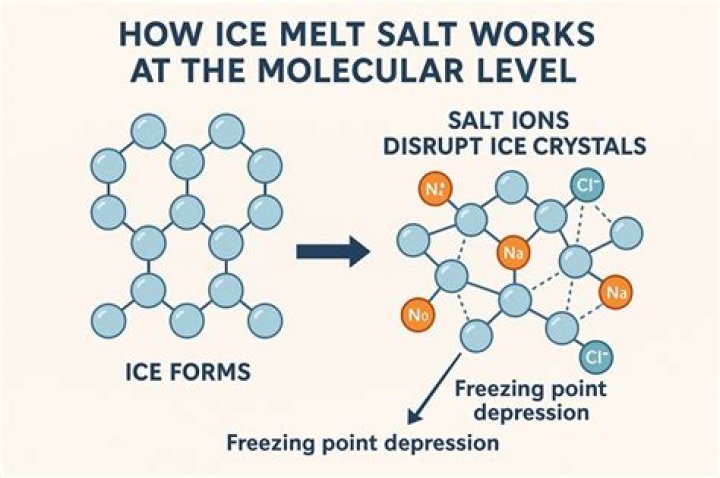
The salt lowers the temperature at which water freezes, so with salt ice will melt even when the temperature is below the normal freezing point of water. Technically, the temperature that the salt lowers is called the freezing point.
How does salt stop ice cream melting?
Similar to sugar, salt affects how water freezes and effectively lowers the freezing/melting point of water. Creating a saltwater slush and packing this around our ice cream base allows us to cool the base enough so that it starts to thicken and freeze before the ice melts completely.
Why is rock salt better for making ice cream?
Adding salt lowers the freezing temperature of the water and for wintery roads, it means that the water won’t freeze as easily. For our ice cream, it allows the temperature of the mixture around the ice cream to get colder. Rock salt isn’t processed as much as table salt, and is much chunkier.
What happens when you add salt to ice cream?
Salt lowers the temperature at which water freezes. So when you mix ice and salt the resulting slushy brine is actually colder than pure ice would be. By surrounding the mixing canister in the ice cream maker with this super cold brine, you bring the temperature of the ice cream mixture down to its freezing point. Ice alone cannot do that.
What happens to the temperature of ice when you add salt?
Salt Lowers the Temperature of Ice Water When you add salt to ice (which always has an outer film of water, so it’s technically ice water), the temperature can drop from freezing or 0 °C to as low as -21 °C. That’s a big difference! Why does the temperature get lower?
Why does Ice Cream Freeze at lower temperature than water?
The next thing to understand is that ice cream freezes (and melts!) at a lower temperature than water. The sugar and fats in the mix interfere with the formation of ice crystals, and it takes a colder temperature to get the ice cream to really freeze.
Why do you have to use ice to make ice cream?
The sugar and fats in the mix interfere with the formation of ice crystals, and it takes a colder temperature to get the ice cream to really freeze. Therefore, we can’t use straight ice to chill the ice cream base, because the ice will melt before the base gets cold enough.
Why does adding salt to ice cream make it colder?
Adding salt lowers the freezing temperature of the water and for wintery roads, it means that the water won’t freeze as easily. For our ice cream, it allows the temperature of the mixture around the ice cream to get colder. Since the ice cream isn’t just water, it needs to be a little below 32°F to freeze.
Salt Lowers the Temperature of Ice Water When you add salt to ice (which always has an outer film of water, so it’s technically ice water), the temperature can drop from freezing or 0 °C to as low as -21 °C. That’s a big difference! Why does the temperature get lower?
The next thing to understand is that ice cream freezes (and melts!) at a lower temperature than water. The sugar and fats in the mix interfere with the formation of ice crystals, and it takes a colder temperature to get the ice cream to really freeze.
When to add rock salt to ice cream?
If, after about 10 minutes, your cream is just starting to firm up, you have a good amount of salt in the ice bath. In addition to the larger grain size and easier control over the freezing process, another benefit of rock salt is that it is cheaper than table salt. Making ice cream can use a great deal of salt.

