Is melting ice cream exothermic?
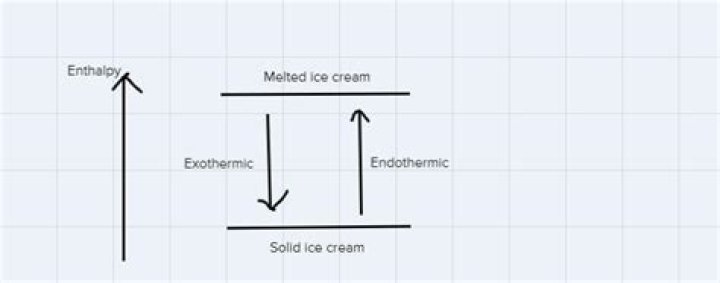
Is melting ice cream exothermic?
Basically, melting ice is an endothermic reaction because the ice absorbs (heat) energy, which causes a change to occur.
What type of energy is a melting ice cream?
ice cream melts on a warm day because heat energy is transferred from the air (because it is at a higher temperature) to the ice cream (because it is at a lower temperature).
When ice cream melts is this an endothermic or exothermic reaction?
Melting of Ice is an Endothermic process ,means that ice takes heat energy from surrounding to change its state from a solid to a liquid. Ice is made up due to Hydrogen bonding between molecules of water (H2O). To break these bonds we require heat energy.
Is making homemade ice cream endothermic or exothermic?
Making ice cream is an endothermic reaction. Our results showed that in order to freeze the ice cream, we would have to make the ice colder. In order to make the ice colder, it must absorb the energy from the warmer ice cream mixture.
Is melting Chocolate exothermic or endothermic?
endothermic – An endothermic reaction occurs when the elements are absorbing the heat allowing the solution to melt and bond together. Also another time where there is a chemical reaction is when the chocolate is melting in your mouth also known as mouthfeel.
Is melting always endothermic?
It requires energy for a solid to melt into a liquid. However, it can be used for both the melting and the solidification processes as long as you keep in mind that melting is always endothermic (so ΔH will be positive), while solidification is always exothermic (so ΔH will be negative).
When ice cream is melting down what type of energy is decreasing?
kinetic energy
For example, as heat transfers into the ice, the ice molecules move faster and eventually the ice melts. Conversely, when heat transfers from your beverage to the ice and the temperature of the liquid drops, the molecules in the beverage slow down. When those molecules slow down, their kinetic energy decreases.
Is melting water exothermic?
Changes of state involve a solid melting, a liquid freezing, a liquid boiling or a gas condensing. When these molecules condense to form liquid water again, the energy put into the system must be released. And this stored energy is let out as exothermic heat.
Is Melting Chocolate exothermic or endothermic?
Is melting copper exothermic?
Is melting copper an example of exothermic? No, heat has to be added to make copper melt. So that’s called an endothermic process. When copper freezes it releases heat, so that’s called exothermic.
How does the exothermic process work in making ice cream?
Endothermic and Exothermic Process in making Ice Cream. Exothermic: Exothermic process happens in making ice cream when the milk freezes, because freezing is one of exothermic process where the heat gets out from the substance to the area, therefore the milk gets cold and it freezes.
Is the melting of ice an exothermic reaction?
Melting ice is endothermic — you can see this by putting a thermometer in a glass of warm water, adding an ice cube, and watching the temperature go down as the ice melts. The melting process needs heat to proceed and takes it from the warm water. Is melting a solid exothermic?
What makes an ice cream spoon melt ice cream?
Its beautiful, sleek design actually serves a purpose and could offer a solution to that no-budge tub. Architect Naoki Terada crafted this spoon entirely from aluminum, a metal high in thermal conductivity. When you hold the spoon from its handle, the heat from your hand is sent up the utensil to the part that makes contact with the ice cream.
How does salt and ice dissolve in ice cream?
This is an endothermic process. The salt and the ice absorb the energy from the milk, which is an endothermic process. It makes the milk releases energy for the salt and ice to dissolve, which is an exothermic process. We shake the container to make the transfer of energy even faster. You are commenting using your WordPress.com account.
How does the exothermic reaction in ice cream work?
Exothermic reaction Ice has to absorb energy in order to melt. When you use ice to cool the ingredients for ice cream, the energy is absorbed from the ingredients and from the outside environment. When you add salt to the ice, it lowers the freezing point of the ice,…
What happens when milk is turned into ice cream?
This ice cream making process involves endothermic and exothermic reaction. Ice cream is a liquid that freezes, and liquid has more energy than solid. In order to freeze the milk into ice cream, the milk needs to lose or ‘release’ energy to change into solid ice cream, when this happens: exothermic reaction occurs.
How is the melting of an Ice Cube an endothermic reaction?
Re: Endothermic Reaction. As such, the heat energy from your hand is transferred to the ice cube, causing it to melt. In other words, heat is being transferred into your system (ice cube) from the surroundings (hand), making the melting of the ice cube endothermic (endothermic roughly meaning heat energy going in (to the system)).
Its beautiful, sleek design actually serves a purpose and could offer a solution to that no-budge tub. Architect Naoki Terada crafted this spoon entirely from aluminum, a metal high in thermal conductivity. When you hold the spoon from its handle, the heat from your hand is sent up the utensil to the part that makes contact with the ice cream.